|
alkali, and alkaline-earth metals, tend to be good reducing agents, as their valence electrons, whose radial orbit DEFINES the atomic radius, tend to be readily oxidized.\) increases slightly the ionization energy decreases.Īs the name suggests, electron affinity is the ability of an atom to accept an electron. On the other hand, the larger elements, i.e. Bohr models for the first three periods of the periodic table are shown below. Learn more about the elements with fully descriptive write-ups and visualizations. They are both related to the fact that as you move from top to bottom, the effective nuclear charge, Z eff, is essentially constant, but the valence electrons are in higher and higher n shells. Discover the interactive periodic table of elements with Ptable, a web-based tool that lets you explore the properties, trends, orbitals, isotopes, and compounds of each element. nitrogen, fluorine, oxygen, TEND to be very powerful oxidants, and this is also manifested by their small atomic size. There are two trends that we are focussing on for periodic trends moving from the top to the bottom on the periodic table in the same group. Excluding the Noble Gases, the smaller atoms from the right hand side, i.e. It follows that the SMALLEST atoms derive the right of the Table as we face it. In the same group, the effective nuclear charge does not change. Of course, the diagram shows NO data (it should do so), but the relative size of the atoms across the Period, and down the Group is clear. When an atom gains or loses a valence electron, it forms an ion. The atomic radius for the halogens increases down the group as n increases. (a) The radius of an atom is defined as one-half the distance between the nuclei in a molecule consisting of two identical atoms joined by a covalent bond. Atomic radius is the distance from the atom’s nucleus to the outer edge of the electron cloud. The trends for the entire periodic table can be seen in Figure 12.1. This decreases the pull on valence electrons towards the nucleus. Electron-electron repulsions: due to their like charges, electron pairs orient themselves as far away as possible from each other, causing the electron cloud to expand (justifies trends across a period). And the best metric that illustrates this trend is the well-known diminution of atomic radii across the Period from left to right? And of course, we should look at some data. The nonmetal atom takes one or more valence electrons from the metal atom. Other trends that are important in understanding effective nuclear charge on the periodic table, or z effective trend. Now it is a fact that the nuclear charge is SHIELDED very poorly by incomplete electronic shells. This instructor led activity will produce a partially filled periodic table that contains electron-dot models for the first twenty elements in the appropriate boxes. Moving down a group, atoms add electron shells so the atomic radius increases and it takes less energy to remove electrons. Metallic character increases moving down a periodic table group and decreases moving across a period. Since metals have few valence electrons, they tend to increase their stability by losing electrons to become cations. However, there are also other patterns in chemical properties on the periodic table. Both metallic and nonmetallic character are periodic table trends. 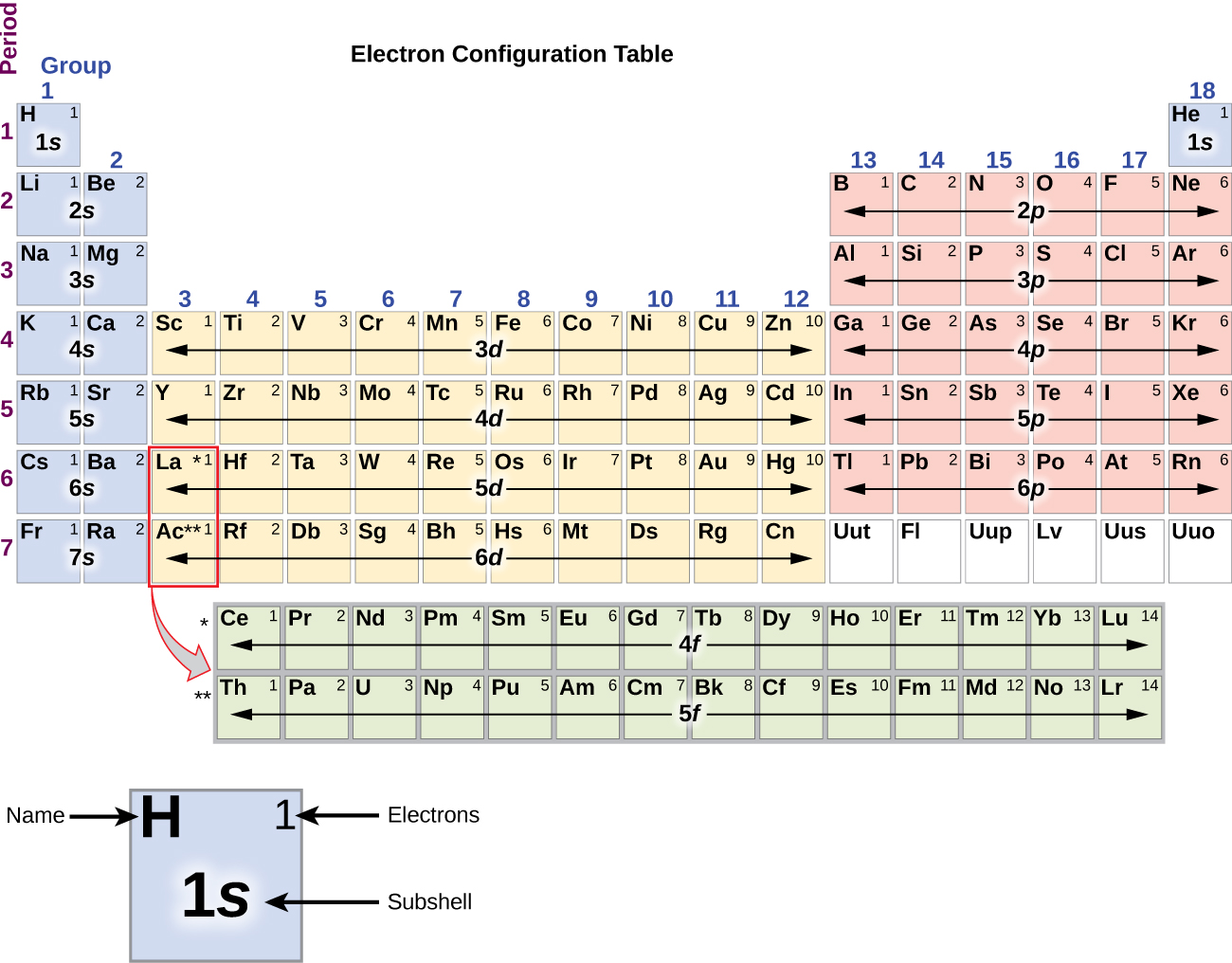
This similarity occurs because the members of a group have the same number and distribution of electrons in their valence shells. 
The chemistry and atomic structure of the elements is a contest between (i) nuclear charge, conveniently represented by #Z_"the atomic number"#, and (ii) shielding by other electrons. The elements in groups (vertical columns) of the periodic table exhibit similar chemical behavior. #"Increase in atomic radii down a Group, a column of the Periodic"#"Table."# And because elements with similar valence electrons will have similar reactivities, they will form similar ions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
